  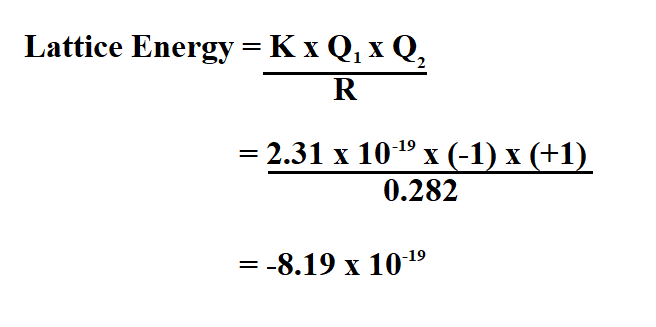
I will explain how you can do this in a moment, but first let's look at how the problem arises. 
In fact, there is a simple way of sorting this out, but many sources don't use it. 
There are two different ways of defining lattice enthalpy which directly contradict each other, and you will find both in common use. Two different ways of defining lattice enthalpy Just don't assume that any bit of data you are given (even by me) is necessarily "right"! It doesn't affect the principles in any way. Values from this now fairly old book often differ slightly from more recent sources.ĭon't worry about this. These came from the Chemistry Data Book edited by Stark and Wallace, published by John Murray. If you use my chemistry calculations book, you will find a slightly different set of numbers. If you are doing a course for 16 - 18 year olds, none of this really matters - you just use the numbers you are given. In the Born-Haber cycles below, I have used numbers which give a consistent answer, but please don't assume that they are necessarily the most accurate ones. Note: While I have been writing this section, the different values for the same piece of data from different data sources has driven me crazy, because there is no easy way of knowing which is the most recent or most accurate data. Unless you go on to do chemistry at degree level, the difference between the two terms isn't likely to worry you. However, the difference is small, and negligible compared with the differing values for lattice enthalpy that you will find from different data sources. In fact, there is a difference between them which relates to the conditions under which they are calculated. Lattice enthalpy and lattice energy are commonly used as if they mean exactly the same thing - you will often find both terms used within the same textbook article or web site, including on university sites. You are right that if you step through what actually happens, you need to know heats of fusion and vaporization as well as heat capacities of all phases for the relevant temperature ranges.This page introduces lattice enthalpies (lattice energies) and Born-Haber cycles. I don't know whether NaCl would go through the liquid state or not, but if it does then we would need to add the latent heat of fusion too.īecause enthalpy is a state function, it does not matter if you choose the actual (realistic) path or take a different path. – Nicolau Saker Neto Jul 8 '14 at 16:39Īccording to the comment above, the heat of vaporization should be about half of the lattice energy (with opposite sign). Thus, it is rather unlikely that, at reasonable temperatures, an ionic gas will break down any further than the smallest possible electrically neutral aggregates. : Roughly, somewhere around half of the lattice enthalpy of a salt comes solely from the binding of the smallest electrically neutral agglomerate that is, it takes about as much energy to break a macroscopic solid NaCl crystal into a gas of ion pairs as it does to break all the ion pairs and create a true plasma. If you look up the atomic distance of $NaCl(g)$, you could estimate the enthalpy of (2) pretty accurately and get a numeric answer. It should be obvious that process (1) cost much more energy than what you get out of process (2) because you are going from 6 nearest neighbors to just one. So to get the required energy, you could first separate all ions (lattice energy) and then calculate the energy of forming individual pairs of sodium and chloride ions (assuming the interaction is purely ionic). If you are interested in this process, you could write it as: what is the product? If we boil sodium chloride at 1 atm pressure, it produces mostly NaCl monomers in the gas phase, see. What are the initial and final temperatures, what is the pressure? What does it mean to vaporize solid NaCl or any ionic compound, i.e. Source: diagram adapted from the cited textbook.īut do we need to add this energy again when we are trying to vaporize an ionic compound? All these processes are experimentally accessible except for the last, so you can determine the lattice energy this way. We can get the same energy in a thought experiment (path 2 in the diagram), first turning all species into gas, then into atoms, then transferring the electron and - finally - forming the lattice from the individual anions and cations. As the diagram below shows, we know the enthalpy of reaction for: This would be difficult to do experimentally, but the value may be determined by using a Born Haber cycle. Here is the textbook explanation ( Fleming: Physical Chemistry): The lattice energy is the energy required to separate the ions in an ionic lattice so that they are at infinite distance (but still ions). Now, according to wikipedia, NaCl has a lattice energy of −756 kJ/mol.įirst, we have to understand the term lattice energy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
